Process
Electrodeposition of Copper Oxide
- Cleaned substrate in ultrasonic bath with IPA and DI water
- Prepared 0.25M CuSO₄ solution; adjusted pH to 10 with NaOH and 3M lactic acid at ~50°C
- Masked substrate edges with Kapton tape to preserve FTO contact
- Deposited at -1.0V for 40 minutes with graphite counter electrode
- Rinsed with DI water and dried
Thermal Oxidation
- Removed Kapton; placed samples in box furnace at 425°C for 1 hour
- Cooled to below 20°C before opening
ZnO Sputtering
- Re-masked edges; used Zn target with pure O₂ as process gas
- Sputtered ~5 minutes
Aluminum Contact Deposition
- Kept masking; used Al target with Ar sputter gas; ~8 minutes
- Final thermal oxidation at 425°C for 1 hour

Microscopy Results
AFM of CuO After Deposition
AFM after CuO deposition showed pronounced surface texturing. Some peaks appeared incomplete, likely due to inspecting the sample mid-deposition.

SEM Results




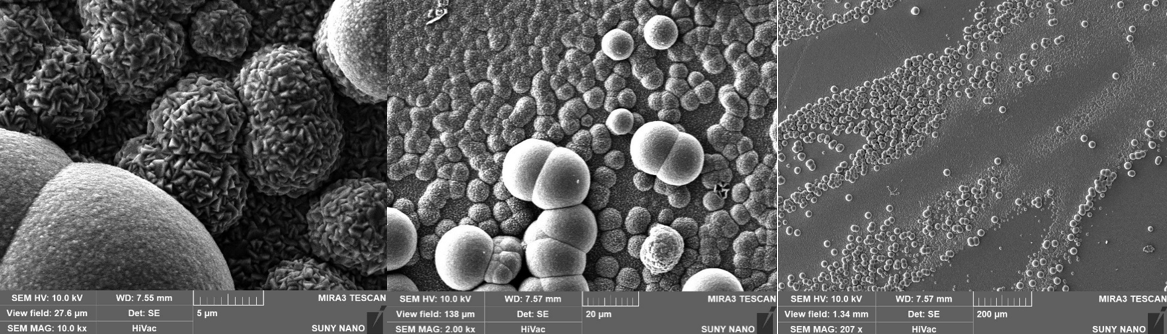
IV Characterization
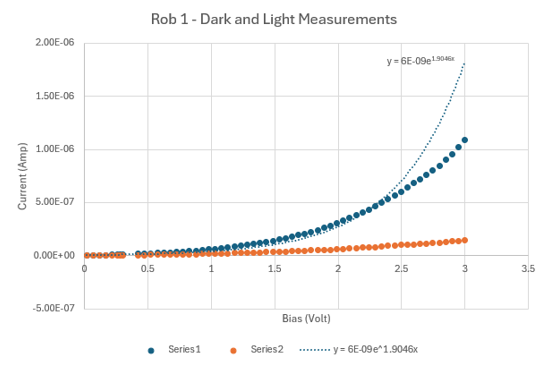
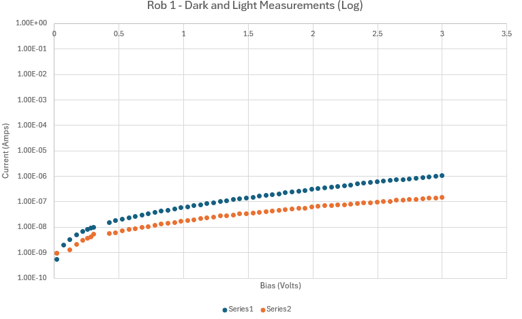
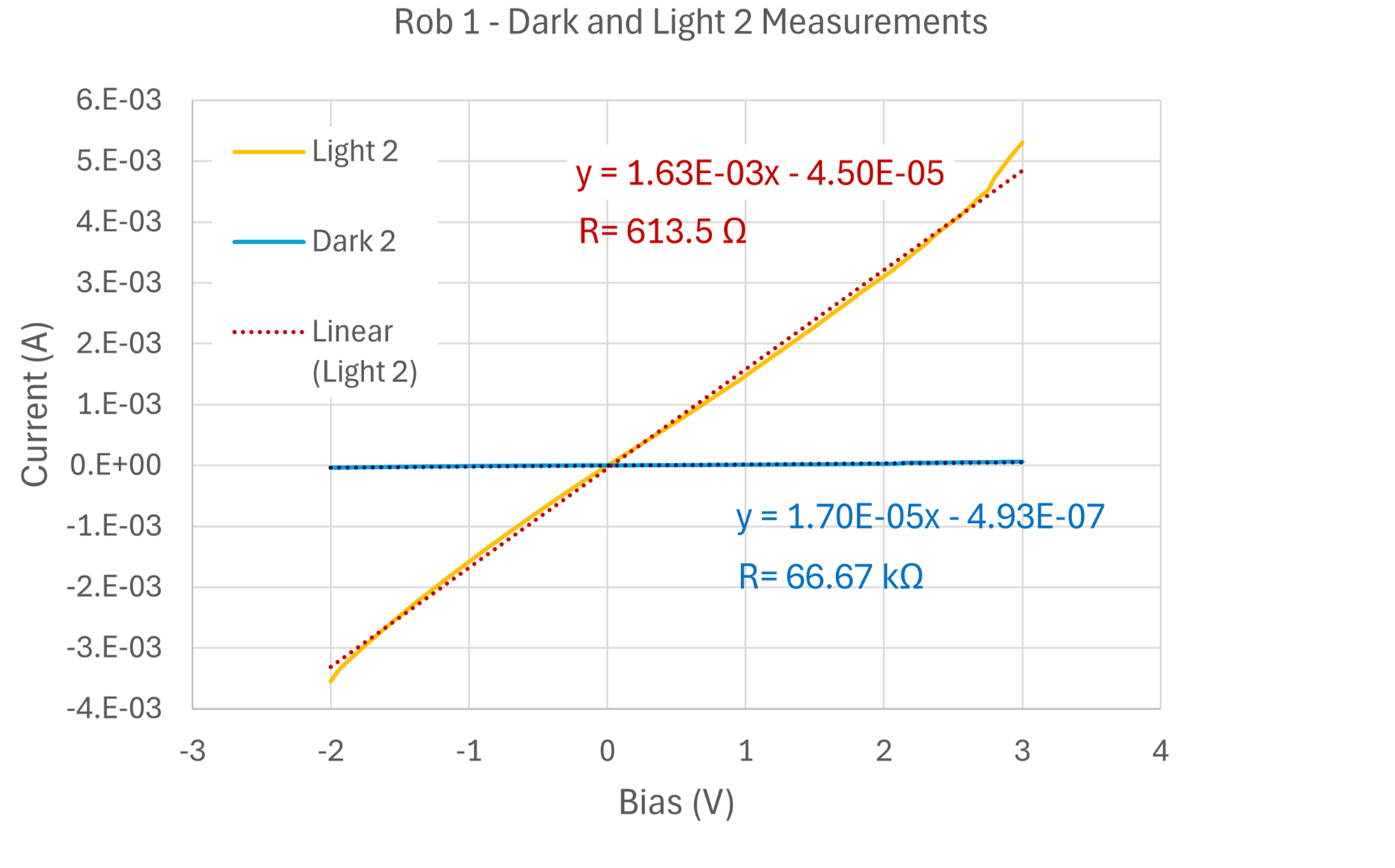
Dark IV confirmed diode behavior. Under illumination, the curves suggested significant recombination losses consistent with the surface defects and non-uniform coverage visible in SEM. Shunt resistance was low, indicating leakage paths likely from the incomplete aluminum coverage and porous CuO layer.
Challenges & Lessons
- Handling samples between furnace steps introduced contamination; cleaning between each step (ultrasonic IPA + DI water) would reduce surface defects
- Loose contact during copper deposition on one sample caused non-uniform current density, visible as discontinuous regions and spherical particles in SEM
- Without introducing O₂ gas flow during oxidation, the growth rate diverged from simulation predictions
Future Work
- Tighter process control at each step, particularly cleaning between depositions
- Introduce proper dry O₂ flow during thermal oxidation steps
- Investigate conformal ZnO deposition to reduce interface defects

